SUSTAINABLE POLYMER AND COLLOID CHEMICAL ENGINEERING
BE PART OF THE SOLUTION
“Environmental sustainability sits at the heart of our chemical engineering approach to innovate in polymer and colloid science.”
PAPERS
We present the latest of our research findings here on this site. For a complete list of our published works see our publications page.

BONLAB BLOG

BONLAB EVENTS
BONLAB ON INSTAGRAM

Liquid Formulations
SUSTAINABILITY & ENVIRONMENT
From developing greener materials and processes to growing more sustainable supply chains, a new £13.6 million research hub, funded by the Engineering and Physical Sciences Research Council (EPSRC), will help researchers at the University of Warwick tackle some of the UK’s biggest manufacturing challenges.
The new Manufacturing Research Hub in Sustainable Engineering Plastics (SEP) will be led by Professor of Polymer Processes, Ton Peijs, at WMG, and has Professor of Polymer and Colloid Chemical Engineering, Stefan Bon, at the Department of Chemistry as one of the co-investigators.
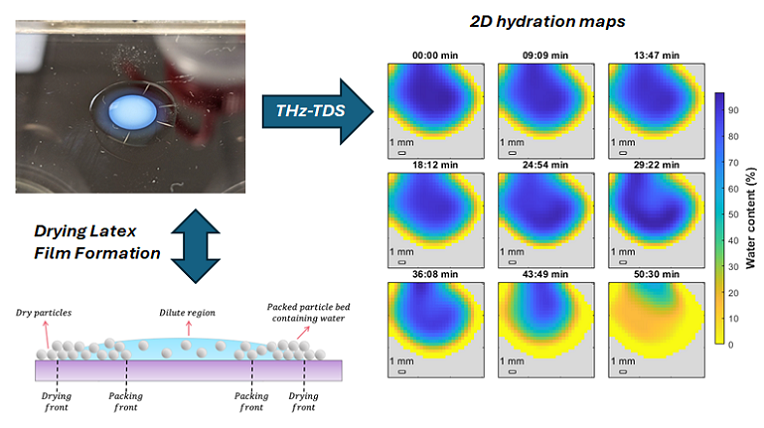
Phase change materials, also known as PCMs, can capture, store, and release energy when they undergo a phase transition. One class of PCMs with relatively high latent energy storage capabilities are alkane-based waxes. These store energy upon melting and will release it again when they solidify through crystallization. To make use of this so-called latent heat is helpful for many applications, and indeed can be found, for example, in building insulation and temperature regulation materials. If, however, we would like to use this concept in a temperature-regulating fluid, we need to disperse the PCMs into a liquid, such as water, that in itself has a high heat capacity and thus the ability to store energy.
Water-based pressure sensitive adhesives (PSAs) are typically made by emulsion polymerization using a low glass transition temperature base monomer, such as n-butyl acrylate or 2-ethyl hexylacrylate, together with a range of functional comonomers. Typically these include a high glass transition temperature comonomer, such as styrene or methyl methacrylate and monomers that can promote wetting and undergo secondary interactions such as (meth)acrylic acid.
Labels are big business. A typical label has multiple layers: a topcoat for protection, the face stock, which contains the message in the form of text and/or images, a pressure-sensitive adhesive, and a release liner, which often has a release coating. The release liner and coating are only there to protect the label from sticking to things you do not wish it would stick to. You remove the liner when you wish to apply the label onto your substrate of choice, for example, a bottle containing a drink.
Imagine a label without a release liner and coating, imagine a label that could be activated at the moment you want it to stick to a substrate, a stick-on-demand linerless label.
BonLab has designed and developed a concept and prototype for a sustainable solution: a mesh reinforced pressure-sensitive adhesive for linerless label design.
A fresh lick of paint breathes new life into a tired looking place. Ever wondered how a thin layer of paint is so effective in hiding what lies underneath from vision? Beside colour pigments, and a binder that makes it stick, paints contain microscopic particles that are great at scattering light and turning that thin layer of paint opaque. The golden standard for these opacifiers is small titanium dioxide particles, of dimensions considerably smaller than one micron. Their use is not without controversy, as they pose a significant environmental burden, with a substantial carbon footprint and a questionable impact on human health. Ideally, though, titanium dioxide should be replaced, but the list of safe, high refractive materials is very limited. Here we provide a potential solution.
A hydrogel is a solid object predominantly composed of water. The water is held together by a cross-linked 3D mesh, which is formed from components such as polymer molecules or colloidal particles. Hydrogels can be found in a wide range of application areas, for example food (think of agar, gelatine, tapioca, alginate containing products), and health (wound dressing, contact lenses, hygiene products, tissue engineering scaffolds, and drug delivery systems).
In Nature hydrogels can be found widely in soft organisms. Jellyfish spring to mind. These are intriguing creatures and form an inspiration for an area called soft robotics, a discipline seek to fabricate soft structures capable of adaptation, ultimately superseding mechanical hard-robots. Hydrogels are an ideal building block for the design of soft robots as their material characteristics can be tailored. It is however, challenging to introduce and program responsive autonomous behaviour and complex functions into man-made hydrogel objects.
Ross Jaggers and prof.dr.ir. Stefan Bon at BonLab have now developed technology that allows for temporal and spatial programming of hydrogel objects, which we made from the biopolymer sodium alginate. Key to its design was the combined use of enzyme and metal-chelation know-how.

A Mechanistic Understanding of
POLYMER AND COLLOID SCIENCE
Single-layer graphene is interesting as a flexible 2D material, with xy-dimensions variable up to a centimetre in length and a z-thickness of a single carbon atom. It conducts heat and electricity, has excellent mechanical strength, and is impermeable to gases except hydrogen gas. Its drawback: how to disperse it in a liquid. When you try to do this flexible sheets of graphene tend to stack as a result of attractive van der Waals interactions, making it virtually an impossible material to disperse as single sheets.
Labels are big business. A typical label has multiple layers: a topcoat for protection, the face stock, which contains the message in the form of text and/or images, a pressure-sensitive adhesive, and a release liner, which often has a release coating. The release liner and coating are only there to protect the label from sticking to things you do not wish it would stick to. You remove the liner when you wish to apply the label onto your substrate of choice, for example, a bottle containing a drink.
Imagine a label without a release liner and coating, imagine a label that could be activated at the moment you want it to stick to a substrate, a stick-on-demand linerless label.
BonLab has designed and developed a concept and prototype for a sustainable solution: a mesh reinforced pressure-sensitive adhesive for linerless label design.
A mini-emulsion polymerization is a variation on the more conventional emulsion polymerization process in that in the ideal scenario latex particles are formed by monomer droplet nucleation. The monomer droplets are turned into polymer particles. The trick to achieve this is to shrink monomer emulsion droplets to sub-micrometer diameters. For this two ingredients are key, one is a lyophobe, a compound that dissolves in the monomer droplet but does not like to partition into the continuous phase, here water. Typically n-hexadecane is used. This compound suppresses coarsening, also called Ostwald ripening, of the droplets by providing an Osmotic counter pressure. The other essential ingredient is a surfactant which aids to stabilize the large combined surface area of the droplets and keeps then from colliding and fusing (colloidal stability).
The use of molecular surfactants, however, can have negative impacts when the polymer latex is used in formulations and applications as the surfactant can migrate. For example in a clear coating it could lead to uptake of water, causing the transparent coating to become opaque, a phenomenon known as water whitening.
When we synthesize polymer colloids by emulsion polymerization, molecular surfactants are often employed. These are required to keep the polymer latex particles dispersed in the water phase, so that they do not clump together, a phenomenon known as coagulation. Keeping polymer dispersions stable is especially important in end applications, such as waterborne coatings and adhesives.
A downside of the use of surfactant molecules is that they can desorb from the surface of the latex particles. This makes the particles colloidally unstable, and they coagulate. This can be disastrous in product formulations, such as water-based paints which have many components. Another downside of this mobility of the surfactant molecules is that they can migrate in the final coating, once applied on a substrate. This leads to deterioration of the properties of the coated film.
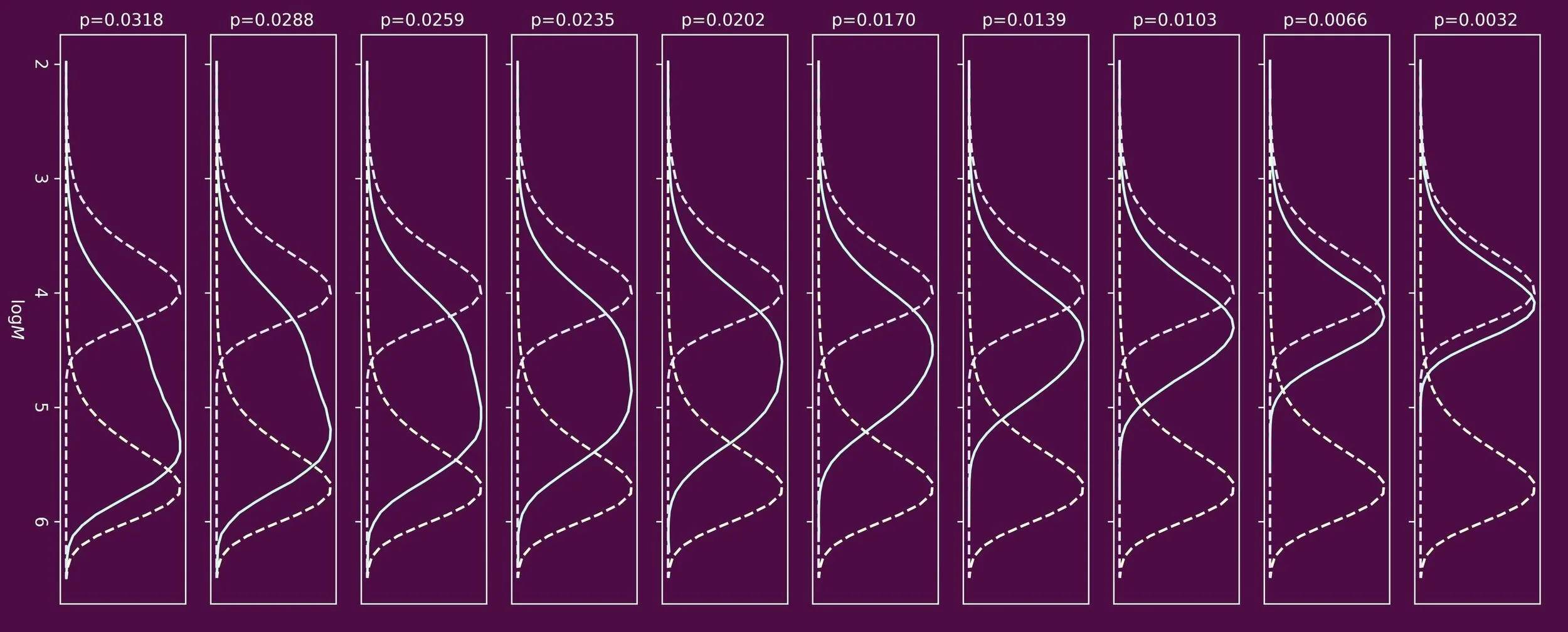
Synthetic polymers in most cases do not have one bespoke molecular weight. A sample typically consists of a large number of individual polymer chains, each having a different molecular weight. The average molecular weights and the shape of the molecular weight distribution are a kinetic fingerprint of how to polymer material was made. The resulting molecular weight distribution dictates physical and mechanical properties.
In free radical polymerizations, four key mechanistic events need to be considered. These are initiation, propagation, termination, and chain transfer. The latter often gets brushed under the carpet in introductory textbooks, but is pivotal.
When one targets polymers of low molecular weight, chain transfer agents are often used. One prominent class of chain transfer agents are thiol compounds, for example n-dodecanethiol. To understand how the molecular weight distribution develops throughout the polymerization process, the ability to determine the reactivity of the chain transfer agent is crucial. This reactivity is often expressed in the form of a chain transfer constant, Ctr, which is the ratio of the rate coefficients of chain transfer and propagation.
Emulsion polymerization is of pivotal importance as a route to the fabrication of water-based synthetic polymer colloids. The product is often referred to as a polymer latex and plays a crucial role in a wide variety of applications spanning coatings (protective/decorative/automotive), adhesives (pressure sensitive/laminating/construction), paper and inks, gloves and condoms, carpets, non-wovens, leather, asphalt paving, redispersible powders, and as plastic material modifiers.
Since its discovery in the 1920s the emulsion polymerization process and its mechanistic understanding has evolved. Our most noticeable past contributions include the first reversible-deactivation nitroxide-mediated radical emulsion polymerization (Macromolecules 1997: DOI 10.1021/ma961003s), and the development and mechanistic understanding of Pickering mini-emulsion (Macromolecules 2005: DOI 10.1021/ma051070z) and emulsion polymerization processes (J. Am. Chem. Soc. 2008: DOI 10.1021/ja807242k). The latest on nano-silica stabilized Pickering Emulsion Polymerization from our lab can be found here.
One quest in emulsion polymerization technology that remains challenging and intriguing is control of the particle morphology. It is of importance as the architecture of the polymer colloid influences its behavioural properties when used in applications. We now report in ACS Nano an elegant innovation in the emulsion polymerization process which makes use of nanogels as stabilizers and allows us to fabricate Janus and patchy polymer colloids.

Reconfigurable Nanoscale Optical and Mechanical Metamaterials (ReNOMMs)
SUPRACOLLOIDAL METAMATERIALS
Researchers from the University of Cambridge and the University of Warwick have secured £2.3M in UKRI funding to create materials with radically new optical and mechanical properties, that can be produced at scale and low cost.
Labels are big business. A typical label has multiple layers: a topcoat for protection, the face stock, which contains the message in the form of text and/or images, a pressure-sensitive adhesive, and a release liner, which often has a release coating. The release liner and coating are only there to protect the label from sticking to things you do not wish it would stick to. You remove the liner when you wish to apply the label onto your substrate of choice, for example, a bottle containing a drink.
Imagine a label without a release liner and coating, imagine a label that could be activated at the moment you want it to stick to a substrate, a stick-on-demand linerless label.
BonLab has designed and developed a concept and prototype for a sustainable solution: a mesh reinforced pressure-sensitive adhesive for linerless label design.
A fresh lick of paint breathes new life into a tired looking place. Ever wondered how a thin layer of paint is so effective in hiding what lies underneath from vision? Beside colour pigments, and a binder that makes it stick, paints contain microscopic particles that are great at scattering light and turning that thin layer of paint opaque. The golden standard for these opacifiers is small titanium dioxide particles, of dimensions considerably smaller than one micron. Their use is not without controversy, as they pose a significant environmental burden, with a substantial carbon footprint and a questionable impact on human health. Ideally, though, titanium dioxide should be replaced, but the list of safe, high refractive materials is very limited. Here we provide a potential solution.

Programmable, Reconfigurable, and Recyclable
DYNAMIC MATERIALS
We set out to develop a prototype for “icy road” warning signs which was able to operate autonomously without the use of electricity, and which could be easily placed onto existing road features, such as street boundary pillars and road safety barriers.
The number of road accidents in the UK under frosty or icy conditions runs in the thousands. Our concept would aid to reduce these numbers, without the introduction of a digital, and thus electric, infrastructure.
The results from our studies are now published open access in the Journal of Materials Chemistry C from the Royal Society of Chemistry. The conceptual road sign application is a multi-lamellar flexible strip.
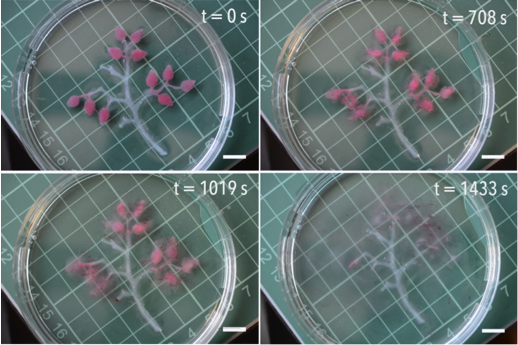
Hydrogels are soft objects that are mainly composed of water. The water is held together by a 3D cross-linked mesh. In our latest work we show that hydrogel beads made from the bio-sustainable polymer alginate can be loaded up with different types of molecules so that the beads can communicate via chemistry.
Chemical communication underpins a plethora of biological functions and behaviours. Plants, animals and insects rely on it for cooperative action, your body uses it to moderate its internal environment and your cells require it to survive.
A key goal of materials science is to mimic this biological behaviour, and synthetic objects that are able to communicate with one another by the sending and receiving of chemical messengers are of great interest at a range of length scales. The most widely explored platform for this kind of communication is between nanoparticles, and to a lesser extent, vesicles, but to date, very little work explores communication between large (millimetre-sized), soft objects, such as hydrogels.
In our work published in the Journal of Materials Chemistry B, we present combinations of large, soft hydrogel objects containing different signalling and receiving molecules, can exchange chemical signals. Beads encapsulating one of three species, namely the enzyme urease, the enzyme inhibitor silver (Ag+), or the Ag+ chelator dithiothreitol (DTT), are shown to interact when placed in contact with one another. By exploiting the interplay between the enzyme, its reversible inhibitor, and this inhibitor’s chelator, we demonstrate a series of ‘conversations’ between the beads.
A hydrogel is a solid object predominantly composed of water. The water is held together by a cross-linked 3D mesh, which is formed from components such as polymer molecules or colloidal particles. Hydrogels can be found in a wide range of application areas, for example food (think of agar, gelatine, tapioca, alginate containing products), and health (wound dressing, contact lenses, hygiene products, tissue engineering scaffolds, and drug delivery systems).
In Nature hydrogels can be found widely in soft organisms. Jellyfish spring to mind. These are intriguing creatures and form an inspiration for an area called soft robotics, a discipline seek to fabricate soft structures capable of adaptation, ultimately superseding mechanical hard-robots. Hydrogels are an ideal building block for the design of soft robots as their material characteristics can be tailored. It is however, challenging to introduce and program responsive autonomous behaviour and complex functions into man-made hydrogel objects.
Ross Jaggers and prof.dr.ir. Stefan Bon at BonLab have now developed technology that allows for temporal and spatial programming of hydrogel objects, which we made from the biopolymer sodium alginate. Key to its design was the combined use of enzyme and metal-chelation know-how.